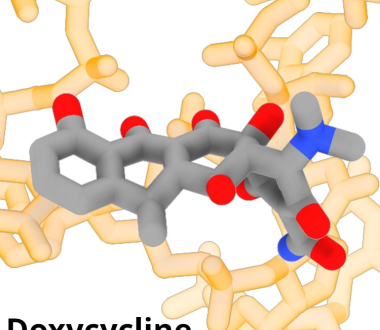
In a study recently published in Biochemistry, Dr. Nilesh Banavali, and colleagues in the Wadsworth Center's Division of Genetics, describe how medically important antibacterials bind to ribosomes of Borrelia burgdorferi, the causative agent of Lyme disease. The team predicted detailed atomic structures and binding affinities for multiple antibacterials interacting with the pathogen’s ribosomal small subunit.
Why this is a breakthrough:
The study provides high-resolution, three-dimensional models showing how small structural differences among antibacterials influence their binding to the ribosome. Notably, the researchers generated ribosome-bound structural predictions for several drugs that previously lacked such data, including doxycycline, a first-line therapy for Lyme disease.
The optimized computational method can also predict how resistance-associated mutations in bacterial ribosomes reduce antibacterial binding. Importantly, the approach is broadly applicable across pathogens and can identify unintended interactions with human ribosomes – an important factor underlying drug toxicity.
These findings support the rational, structure-based design of improved antibacterials that minimize off-target effects on human cells while maintaining or enhancing activity against specific pathogens. This work highlights the potential for developing safer, narrow-spectrum antibiotics that preserve the human microbiome while effectively treating infection.
Overall, this research underscores New York State’s leadership in advancing innovative approaches to combat antibiotic resistance and infectious diseases.