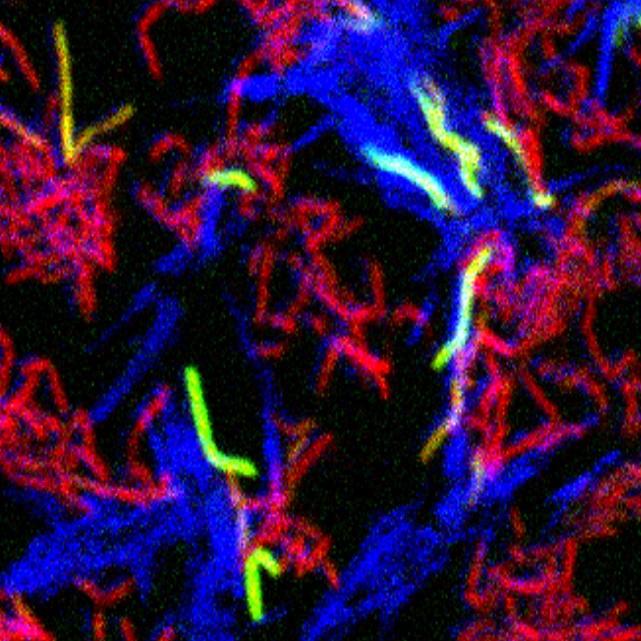
Red and blue cells are non-kin. When they contact each other, red cells turn on a green gene, indicating DCT is in progress. https://pubmed.ncbi.nlm.nih.gov/29941598/
Before COVID most of us probably never thought about how the organisms that cause disease inherit their genetic material and how that impacts their ability to be “successful” pathogens.
Drs. Todd Gray and Keith Derbyshire at the Wadsworth Center have been thinking about this for a long time.
Impact
They study mycobacteria. And while mycobacteria evolve very differently than coronaviruses, they are successful pathogens, and a major public health threat. In fact, one species, Mycobacterium tuberculosis, which causes TB, kills more than 1.5 million people each year, reclaiming its top spot as the most lethal infectious agent as COVID recedes. In addition, related mycobacteria that cause non-tuberculous disease are an increasing health threat.
All Mixed Up
One method that bacteria use to acquire genetic material from a neighbor is called conjugation. The Derbyshire-Gray Laboratory has described a unique type of conjugation in mycobacteria, termed Distributive Conjugal Transfer (DCT). DCT can mix the genomes of two stains of Mycobacterium smegmatis almost as completely as sexual reproduction in animals. DCT begins with direct contact between genetically distinct donor and recipient mycobacterial cells. Donor and recipient cells have a way of distinguishing each other upon contact. If they determine they are in contact with related cells, “kin”, the DCT process is not initiated and genetic transfer does not occur. Thus, transfer of genetic material only occurs between non-kin cells, avoiding nonproductive “inbreeding".
What’s Next?
How can two bacterial cells recognize each other for conjugation? Is there communication between them? The Derbyshire-Gray Laboratory recently identified a cluster of highly variable genes involved in expressing self (kin)-identity. Now they are working to characterize kin recognition in mycobacteria and to determine how it triggers, or blocks, the cell-to-cell communication that leads to DCT.
The five-year award will support work further describing the genes and encoded proteins responsible for discriminating between kin and non-kin mycobacteria. Learning more about DCT will also help model how mycobacteria communicate with one another and how mycobacteria have evolved or continue to evolve.
Project number R01AI181948-01 funded by the National Institute of Allergy and Infectious Diseases. Contents are solely the responsibility of the authors and do not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases.
Related publications
Clark RR, Lapierre P, Lasek-Nesselquist E, Gray TA, Derbyshire KM. A Polymorphic Gene within the Mycobacterium smegmatis esx1 Locus Determines Mycobacterial Self-Identity and Conjugal Compatibility. mBio. 2022 Apr 26;13(2):e0021322. doi: 10.1128/mbio.00213-22. Epub 2022 Mar 17. PMID: 35297678; PMCID: PMC9040860.
Gray TA, Derbyshire KM. Blending genomes: distributive conjugal transfer in mycobacteria, a sexier form of HGT. Mol Microbiol. 2018 Jun;108(6):601-613. doi: 10.1111/mmi.13971. Epub 2018 May 11. PMID: 29669186; PMCID: PMC5997560.
Gray TA, Krywy JA, Harold J, Palumbo MJ, Derbyshire KM. Distributive conjugal transfer in mycobacteria generates progeny with meiotic-like genome-wide mosaicism, allowing mapping of a mating identity locus. PLoS Biol. 2013 Jul;11(7):e1001602. doi: 10.1371/journal.pbio.1001602. Epub 2013 Jul 9. PMID: 23874149